[Thu. 23 Nov. 2017] Prof. Sung-Gon Kim/Department of Chemistry, Kyonggi University, Suwon, Korea
| Speaker | Prof. Sung-Gon Kim |
|---|---|
| Date | Thu. 23 Nov. 2017 |
| Time | 5:00pm |
| Venue | #331, Asan Hall, College of Science |
“Organocatalytic Cascade Reactions for the Asymmetric Synthesis
of Fused-ring Compounds”
Fused-ring structural units including chroman, indoline, pyrrolidine and hydroqunoline are widely distributed in nature and many biologically active compounds. Molecules containing these structural units exhibit a broad range of bioactivities such as anticancer, antiviral, antitumar, antimicrobial, sex pheromone, and central nervous system activity. Owing to the importance of these class of compounds, the stereoselective synthesis of fused-ring compounds is a noteworthy synthetic goal. In this presentation, we demonstrate the asymmetric synthesis of hydroquinoline, hydroquinazoline, and indolines via catalytic cascade reaction.
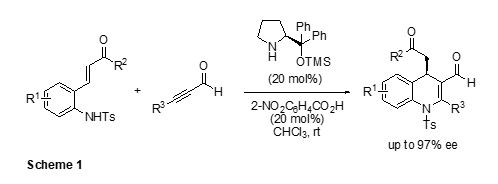
First, A synthetic method for the construction of fully substituted enantioenriched 1,4-dihydroquinolines using an organocatalytic aza-Michael/Michael cascade reaction has been developed (Sheme 1). 1 The asymmetric reaction of 2-(tosylamino)phenyl α,β-unsaturated ketones with alkynyl aldehydes, promoted by diphenylprolinol O-TMS ether as an organocatalyst, generated chiral 1,4-dihydroquinolines in good to high yields with excellent enantioselectivities (up to 97% ee)
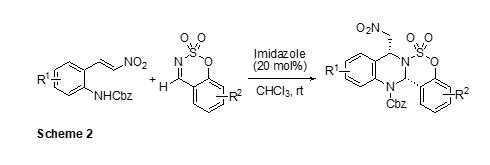
We have also found a concise synthetic route to valuable highly functionalized benzosulfamidate-fused tetrahydroquinazoline (Scheme 2).2 The [4+2] cycloaddition of o-N-Cbz-amino-b-nitrostyrene with benzoxathiazine 2,2-dioxide using an imidazole as the catalyst afforded tetrahydroquinazolines with high diastereoselectivities.
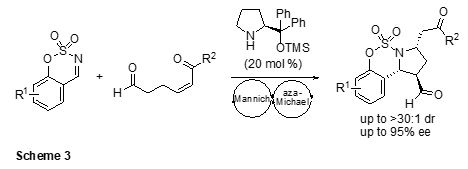
In addition, we have established the synthesis of chiral benzosulfamidate-fused pyrrolidines through an Mannich/aza-Michael cascade reaction of δ-formyl-a,b-unsaturated ketones with cyclic N-sulfimines (Scheme 3).3 This simple domino process afforded diverse highly functionalized pyrrolidines in good yields with high diastero- and enantioselectivities.
Reference
1. Lee, Y.; Heo, S.; Kim, S.-G. Adv. Synth. Catal. 2015, 357, 1545-1550.
2. Sim, J.-T.; Kim, H. Kim, S.-G. Tetrahedron Lett. 2016, 57, 5907-5910.
3. Kim, H.; Kim, Y.; Kim, S.-G. J. Org. Chem. 2017, 82, 8179-8185.
Articles
-
 [Thu. 23 Nov. 2017] Prof. Sung-Gon Kim/Department of Chemistry, Kyonggi Universi...
[Thu. 23 Nov. 2017] Prof. Sung-Gon Kim/Department of Chemistry, Kyonggi Universi...
- [November 14, 2017] Zero-Point Energy, Fluctuations, and Dissipation : A (Mostly)...
- [Thu. 16 Nov. 2017] Prof. Hyungjun Kim/Department of Chemistry, and Graduate scho...
- [Fri. 30 Oct. 2017] Prof. Igor Rodin/Department of Chemistry, Moscow State Univer...
- [October 31, 2017] New ways of vision: Protein structures in translational medici...
- [Thu. 26 Oct. 2017] Dr. Jung-Nyoung Heo/Center for Medicinal Chemistry Research, ...
- [October 25, 2017] Ultrafast electron microscopy: applications in chemical and ma...
- [Mon. 16 Oct. 2017] Prof. Kirk S. Schanze/Department of Chemistry, University of ...
- [Fri. 13 Oct. 2017] Dr. J. Nathan Hohman/The Molecular Foundry, Lawrence Berkeley...
- [Thu. 12 Oct. 2017] Student Host Colloquium : Versatile Dendrimer-Encapsulated Na...
-
 [October 10, 2017] Novel Terahertz Spectroscopies
[October 10, 2017] Novel Terahertz Spectroscopies
- [Thu. 28 Sep. 2017] Prof. Eunsung Lee/Department of Chemistry, POSTECH and Center...
- [Thu. 14 Sep. 2017] Prof. Dokyoung Kim/Department of Anatomy and Neurobiology, Ky...
-
 Fall semester 2017 Seminar Schedule
Fall semester 2017 Seminar Schedule
-
 7th Jilin-Korea-Waseda Alliance Annual Symposium
7th Jilin-Korea-Waseda Alliance Annual Symposium
- [Wed. 5 July. 2017] Seminar is canceled!
- [Fri. 16 Jun. 2017] Prof. Martin Thuo/Department of Materials Science and Enginee...
- [Thu. 8 Jun. 2017] Prof. Won-jin Chung/Department of Chemistry, GIST, Gwangju, Re...
- [Thu. 1 Jun. 2017] Prof. Jong-Man Kim/Department of Chemical Engineering, Hanyang...
- [Thu. 25 May. 2017] Prof. Wonsuk Kim/Department of Chemistry and Nano Science, Ew...
Designed by sketchbooks.co.kr / sketchbook5 board skin