Decoding the Roles of Amyloid-β (1-42)'s Key Oligomerization Domains toward Designing Epitope-Specific Aggregation Inhibitors
| author | Hugh I. Kim |
|---|---|
| Homepage | https://www.hughkimlab.com/ |
| journal | JACS Au |
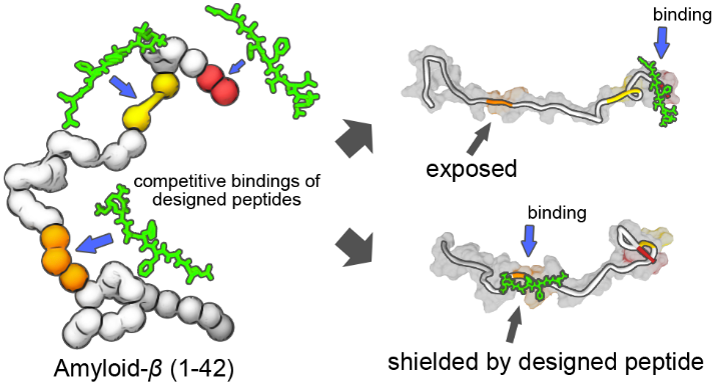
Fibrillar amyloid aggregates are the pathological hallmarks of multiple neurodegenerative diseases. The amyloid-β (1-42) protein, in particular, is a major component of senile plaques in the brains of patients with Alzheimer’s disease and a primary target for disease treatment. Determining the essential domains of amyloid-β (1-42) that facilitate its oligomerization is critical for the development of aggregation inhibitors as potential therapeutic agents. In this study, we identified three key hydrophobic sites (17LVF19, 32IGL34, and 41IA42) on amyloid-β (1-42) and investigated their involvement in the self-assembly process of the protein. Based on these findings, we designed candidate inhibitor peptides of amyloid-β (1-42) aggregation. Using the designed peptides, we characterized the roles of the three hydrophobic regions during amyloid-β (1-42) fibrillar aggregation and monitored the consequent effects on its aggregation property and structural conversion. Furthermore, we used an amyloid-β (1-42) double point mutant (I41N/A42N) to examine the interactions between the two C-terminal end residues with the two hydrophobic regions, and their roles in amyloid self-assembly. Our results indicate that interchain interactions in the central hydrophobic region (17LVF19) of amyloid-β (1-42) are important for fibrillar aggregation, and its interaction with other domains is associated with the accessibility of the central hydrophobic region for initiating the oligomerization process. Our study provides mechanistic insights into the self-assembly of amyloid-β (1-42) and highlights key structural domains that facilitate this process. Our results can be further applied toward improving the rational design of candidate amyloid-β (1-42) aggregation inhibitors.
https://pubs.acs.org/doi/10.1021/jacsau.2c00668
« Prev Resonant Raman-Active Polymer Dot Barcodes for Multiplex Cell...
 Resonant Raman-Active Polymer Dot Barcodes for Multiplex Cell...
2023.05.08by webmaster2
〈
Resonant Raman-Active Polymer Dot Barcodes for Multiplex Cell...
2023.05.08by webmaster2
〈
Flattening bent Janus nanodiscs expands lattice parameters Next »
 Flattening bent Janus nanodiscs expands lattice parameters
2023.05.08by webmaster2
〉
Flattening bent Janus nanodiscs expands lattice parameters
2023.05.08by webmaster2
〉
Articles
- Self-Aggregating Tau Fragments Recapitulate Pathologic Phenotypes and Neurotoxici...
-
 Ultranarrow Mid-infrared Quantum Plasmon Resonance of Self-Doped Silver Selenide ...
Ultranarrow Mid-infrared Quantum Plasmon Resonance of Self-Doped Silver Selenide ...
-
 Molecular Thermoelectricity in EGaIn-Based Molecular Junctions
Molecular Thermoelectricity in EGaIn-Based Molecular Junctions
-
 Perovskite Nanocatalsts Protected by Hermetically Sealing for Highly Bright and S...
Perovskite Nanocatalsts Protected by Hermetically Sealing for Highly Bright and S...
-
 The importance of a charge transfer descriptor for screening potential CO2 reduct...
The importance of a charge transfer descriptor for screening potential CO2 reduct...
-
 Resonant Raman-Active Polymer Dot Barcodes for Multiplex Cell Mapping
Resonant Raman-Active Polymer Dot Barcodes for Multiplex Cell Mapping
-
 Decoding the Roles of Amyloid-β (1-42)'s Key Oligomerization Domains toward Desig...
Decoding the Roles of Amyloid-β (1-42)'s Key Oligomerization Domains toward Desig...
-
 Flattening bent Janus nanodiscs expands lattice parameters
Flattening bent Janus nanodiscs expands lattice parameters
-
 Directing the surface atomic geometry on copper sulfide for enhanced electrochemi...
Directing the surface atomic geometry on copper sulfide for enhanced electrochemi...
-
 High Seebeck Coefficient Achieved by Multinuclear Organometallic Molecular Junctions
High Seebeck Coefficient Achieved by Multinuclear Organometallic Molecular Junctions
-
 Thermopower in Transition from Tunneling to Hopping
Thermopower in Transition from Tunneling to Hopping
-
 An Activity-Based Fluorescent Probe for Imaging Fluctuations of Peroxynitrite (ON...
An Activity-Based Fluorescent Probe for Imaging Fluctuations of Peroxynitrite (ON...
-
 Deep learning for development of organic optoelectronic devices: Efficient prescr...
Deep learning for development of organic optoelectronic devices: Efficient prescr...
-
 Photocatalytic Superoxide Radical Generator that Induces Pytoptosis in Cancer Cells
Photocatalytic Superoxide Radical Generator that Induces Pytoptosis in Cancer Cells
-
 Direct C-H metallation of tetrahydrofuran and application in flow
Direct C-H metallation of tetrahydrofuran and application in flow
-
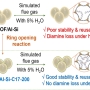 Functionalization of Diamine-Appended MOF-Based Adsorbents by Ring Opening of Epo...
Functionalization of Diamine-Appended MOF-Based Adsorbents by Ring Opening of Epo...
-
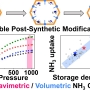 High Gravimetric and Volumetric Ammonia Capacities in Robust Metal-Organic Framew...
High Gravimetric and Volumetric Ammonia Capacities in Robust Metal-Organic Framew...
-
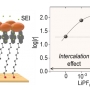 Li-ion Intercalation, Rectification, and Solid Electrolyte Interphase in Molecula...
Li-ion Intercalation, Rectification, and Solid Electrolyte Interphase in Molecula...
-
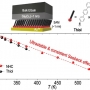 Thermopower of Molecular Junction in Harsh Thermal Environments
Thermopower of Molecular Junction in Harsh Thermal Environments
-
 Microfluidics-Assisted Synthesis of Hierarchical Cu2O Nanocrystal as C2-Selective...
Microfluidics-Assisted Synthesis of Hierarchical Cu2O Nanocrystal as C2-Selective...
Designed by sketchbooks.co.kr / sketchbook5 board skin