Supramolecular Modulation of Structural Polymorphism in Pathogenic α-Synuclein Fibrils Using Cu(II) Coordination
| author | Hugh I. Kim |
|---|---|
| Homepage | http://Angew. Chem. Int. Ed. 2018 (DOI: ...201712286) |
| journal | https://sites.google.com/site/hughkimgroup/ |
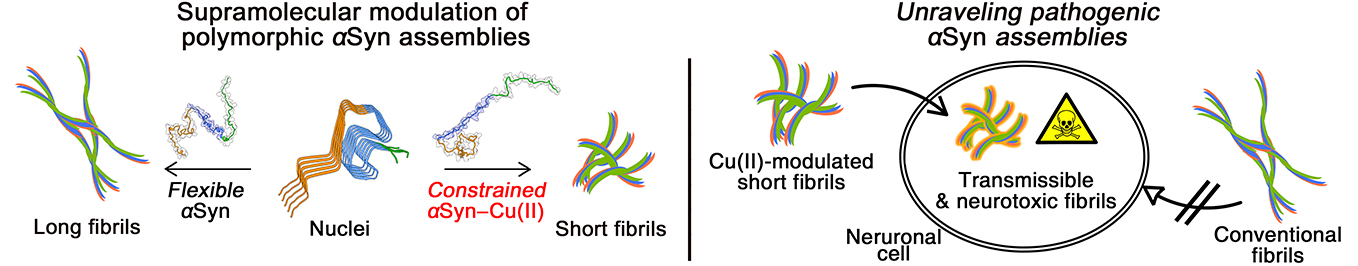
Structural variation of α-synuclein (αSyn) fibrils has been linked to the diverse etiologies of synucleinopathies. However, little is known about what specific mechanism provides αSyn fibrils with pathologic features. Herein, we demonstrate Cu(II)-based supra-molecular approach for unraveling the formation process of pathogenic αSyn fibrils and its application in a neurotoxic mechanism study. The conformation of αSyn monomer was strained by macrochelation with Cu(II), thereby disrupting the fibril elongation while promoting its nucleation. This non-canonical process formed shortened, β-sheet enriched αSyn fibrils (< 0.2 µm) that were rapidly transmitted and accumulated to neuronal cells, causing neuronal cell death, in sharp contrast to typical αSyn fibrils (~1 µm). Our approach provided the supramolecular basis for the formation of pathogenic fibrils through physiological factors such as brain Cu(II).
http://onlinelibrary.
« Prev Molecular Role of Ca2+ and Hard Divalent Metal Cations on Acc...
 Molecular Role of Ca2+ and Hard Divalent Metal Cations on Acc...
2018.01.31by Manager
〈
Molecular Role of Ca2+ and Hard Divalent Metal Cations on Acc...
2018.01.31by Manager
〈
Recent Progress in the Chemistry of Pyridazinones for Functio... Next »
 Recent Progress in the Chemistry of Pyridazinones for Functio...
2018.01.31by Manager
〉
Recent Progress in the Chemistry of Pyridazinones for Functio...
2018.01.31by Manager
〉
Articles
-
 Molecular Role of Ca2+ and Hard Divalent Metal Cations on Accelerated Fibrillatio...
Molecular Role of Ca2+ and Hard Divalent Metal Cations on Accelerated Fibrillatio...
-
 Supramolecular Modulation of Structural Polymorphism in Pathogenic α-Synuclein Fi...
Supramolecular Modulation of Structural Polymorphism in Pathogenic α-Synuclein Fi...
-
 Recent Progress in the Chemistry of Pyridazinones for Functional Group Transforma...
Recent Progress in the Chemistry of Pyridazinones for Functional Group Transforma...
-
 Maskless Arbitrary Writing of Molecular Tunnel Junctions
Maskless Arbitrary Writing of Molecular Tunnel Junctions
-
 Single Component Organic Solar Cells Based on Oligothiophene-Fullerene Conjugate
Single Component Organic Solar Cells Based on Oligothiophene-Fullerene Conjugate
-
 Lanthanide metal-assisted synthesis of rhombic dodecahedral MNi (M=Ir and Pt) nan...
Lanthanide metal-assisted synthesis of rhombic dodecahedral MNi (M=Ir and Pt) nan...
-
 Radially Phase Segregated PtCu@PtCuNi Dendrite@Frame Nanocatalyst for the Oxygen ...
Radially Phase Segregated PtCu@PtCuNi Dendrite@Frame Nanocatalyst for the Oxygen ...
-
 Collision Cross Sections and Ion Structures: Development of a General Calculation...
Collision Cross Sections and Ion Structures: Development of a General Calculation...
-
 Previous Article Next Article Table of Contents Total Syntheses of Arcyriaflavin ...
Previous Article Next Article Table of Contents Total Syntheses of Arcyriaflavin ...
-
 Synthesis, Characterization, and Efficient Catalytic Activities of a Nickel(II) P...
Synthesis, Characterization, and Efficient Catalytic Activities of a Nickel(II) P...
-
 Molecular Insights into Human Serum Albumin as a Receptor of Amyloid-β in the Ext...
Molecular Insights into Human Serum Albumin as a Receptor of Amyloid-β in the Ext...
-
 Two Regioisomeric π-Conjugated Small Molecules: Synthesis, Photophysical, Packing...
Two Regioisomeric π-Conjugated Small Molecules: Synthesis, Photophysical, Packing...
-
 Major Electronic Transition Shift from Bandgap to Localized Surface Plasmon Reson...
Major Electronic Transition Shift from Bandgap to Localized Surface Plasmon Reson...
-
 Rational Design of In Vivo Tau Tangle-Selective Near Infrared Fluorophores: Expan...
Rational Design of In Vivo Tau Tangle-Selective Near Infrared Fluorophores: Expan...
-
 Achieving Highly Efficient Nonfullerene Organic Solar Cells with Improved Intermo...
Achieving Highly Efficient Nonfullerene Organic Solar Cells with Improved Intermo...
-
 Ionic effect on the excited-state proton transfer reactions in aqueous solutions
Ionic effect on the excited-state proton transfer reactions in aqueous solutions
-
 A conductive porous organic polymer with superprotonic conductivity of a Nafion-t...
A conductive porous organic polymer with superprotonic conductivity of a Nafion-t...
-
 Quantum optical measurements with undetected photons through vacuum field indisti...
Quantum optical measurements with undetected photons through vacuum field indisti...
-
 Nanoscale Control of Amyloid Self-Assembly Using Protein Phase Transfer by Host-G...
Nanoscale Control of Amyloid Self-Assembly Using Protein Phase Transfer by Host-G...
-
 A Mitochondria-targeted Cryptocyanine-Based Photothermogenic Photosensitizer
A Mitochondria-targeted Cryptocyanine-Based Photothermogenic Photosensitizer
Designed by sketchbooks.co.kr / sketchbook5 board skin