Molecular Design of Highly Efficient Heavy-Atom-Free Triplet BODIPY Derivatives for Photodynamic Therapy and Bioimaging
| author | Sungnam Park |
|---|---|
| Homepage | https://ultrafastspec.wixsite.com/spark |
| journal | Angewandte Chemie International Edition |
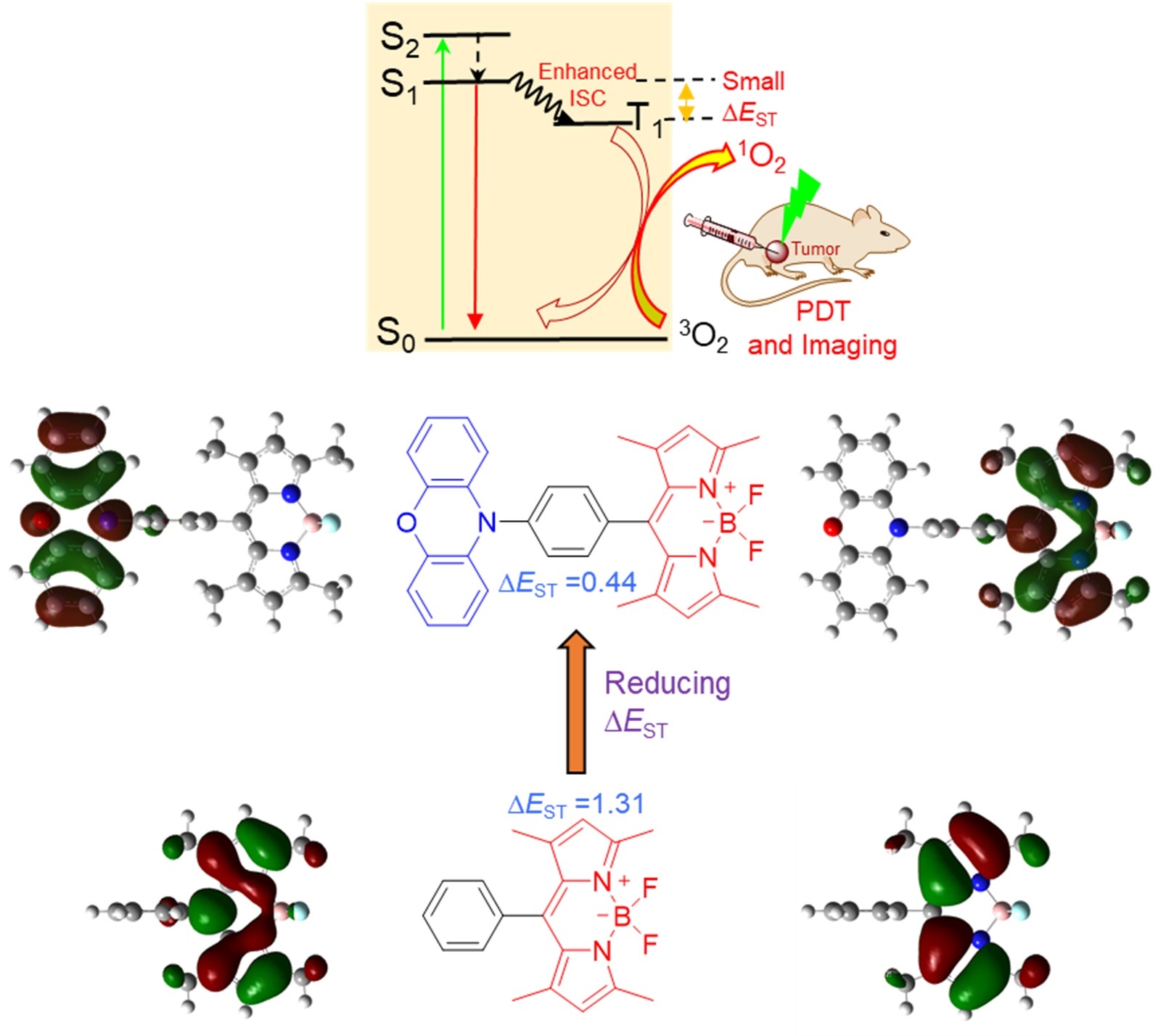
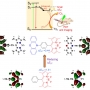
Novel BODIPY photosensitizers were developed for imaging‐guided photodynamic therapy. The introduction of a strong electron donor to the BODIPY core through a phenyl linker combined with the twisted arrangement between the donor and the BODIPY acceptor is essential for reducing the energy gap between the lowest singlet excited state and the lowest triplet state (ΔE ST), leading to a significant enhancement in the intersystem crossing (ISC) of the BODIPYs. Remarkably, the BDP‐5 with the smallest ΔE ST (ca. 0.44 eV) exhibited excellent singlet oxygen generation capabilities in both organic and aqueous solutions. BDP‐5 also displayed bright emission in the far‐red/near‐infrared region in the condensed states. More importantly, both in vitro and in vivo studies demonstrated that BDP‐5 NPs displayed a high potential for photodynamic cancer therapy and bioimaging.
https://doi.org/10.1002/anie.202002843

Sangin Kim (first author, master's course)
« Prev Highly Crystalline Hollow Toroidal Copper Phosphosulfide via ...
 Highly Crystalline Hollow Toroidal Copper Phosphosulfide via ...
2023.05.03by webmaster2
〈
Highly Crystalline Hollow Toroidal Copper Phosphosulfide via ...
2023.05.03by webmaster2
〈
Transformation of Colloidal Quantum Dot: From Intraband Trans... Next »
 Transformation of Colloidal Quantum Dot: From Intraband Trans...
2023.05.03by webmaster2
〉
Transformation of Colloidal Quantum Dot: From Intraband Trans...
2023.05.03by webmaster2
〉
Articles
-
 Synergistic Effects on Lithium Metal Batteries by Preferential Ionic Interactions...
Synergistic Effects on Lithium Metal Batteries by Preferential Ionic Interactions...
-
 Operando Raman and UV-Vis spectroscopic investigation of the coloring and bleachi...
Operando Raman and UV-Vis spectroscopic investigation of the coloring and bleachi...
-
 Ultrafast intraband Auger process in self-doped colloidal quantum dots
Ultrafast intraband Auger process in self-doped colloidal quantum dots
-
 Two-dimensional electronic spectroscopy of bacteriochlorophyll a with synchronize...
Two-dimensional electronic spectroscopy of bacteriochlorophyll a with synchronize...
-
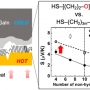 Superexchange Coupling-Induced Enhancements of Thermoelectric Performance in Satu...
Superexchange Coupling-Induced Enhancements of Thermoelectric Performance in Satu...
-
 Fluorescent Diagnostic Probes in Neurodegenerative Diseases
Fluorescent Diagnostic Probes in Neurodegenerative Diseases
- An Ethacrynic Acid-Brominaed BODIPY Photosensitizer (EA-BPS) Construct Enhances t...
-
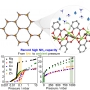 High Ammonia Uptake of a Metal-Organic Framework Adsorbent in a Wide Pressure Range
High Ammonia Uptake of a Metal-Organic Framework Adsorbent in a Wide Pressure Range
-
 Emerging Porous Materials and their Composites for NH3 Gas Removal
Emerging Porous Materials and their Composites for NH3 Gas Removal
-
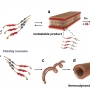 Light-directed trapping of metastable intermediates in a self-assembly process
Light-directed trapping of metastable intermediates in a self-assembly process
-
 Multiply Charged Conjugated Polyelectrolytes as a Multifunctional Inerlayer for E...
Multiply Charged Conjugated Polyelectrolytes as a Multifunctional Inerlayer for E...
-
 Green-, Red-, and Near-Infrared-Emitting Polymer Dot Probes for Simultaneous Mult...
Green-, Red-, and Near-Infrared-Emitting Polymer Dot Probes for Simultaneous Mult...
-
 Intermetallic PtCu Nanoframes as Efficient Oxygen Reduction Electrocatalysts
Intermetallic PtCu Nanoframes as Efficient Oxygen Reduction Electrocatalysts
-
 Pt Dopant: Controlling the IR Oxidation States toward Efficient and Durable Oxyge...
Pt Dopant: Controlling the IR Oxidation States toward Efficient and Durable Oxyge...
-
 Color-Runable Boron-Based Emitters Exhibiting Aggregation-Induced Emission and Th...
Color-Runable Boron-Based Emitters Exhibiting Aggregation-Induced Emission and Th...
-
 Catalytic Nanoframes and Beyond
Catalytic Nanoframes and Beyond
-
 Highly Crystalline Hollow Toroidal Copper Phosphosulfide via Anion Exchange: A Ve...
Highly Crystalline Hollow Toroidal Copper Phosphosulfide via Anion Exchange: A Ve...
-
 Molecular Design of Highly Efficient Heavy-Atom-Free Triplet BODIPY Derivatives f...
Molecular Design of Highly Efficient Heavy-Atom-Free Triplet BODIPY Derivatives f...
-
 Transformation of Colloidal Quantum Dot: From Intraband Transition to Localized S...
Transformation of Colloidal Quantum Dot: From Intraband Transition to Localized S...
-
 Mitochondrial Relocation of a Common Synthetic Antibiotic: A Non-genotoxic Approa...
Mitochondrial Relocation of a Common Synthetic Antibiotic: A Non-genotoxic Approa...
Designed by sketchbooks.co.kr / sketchbook5 board skin