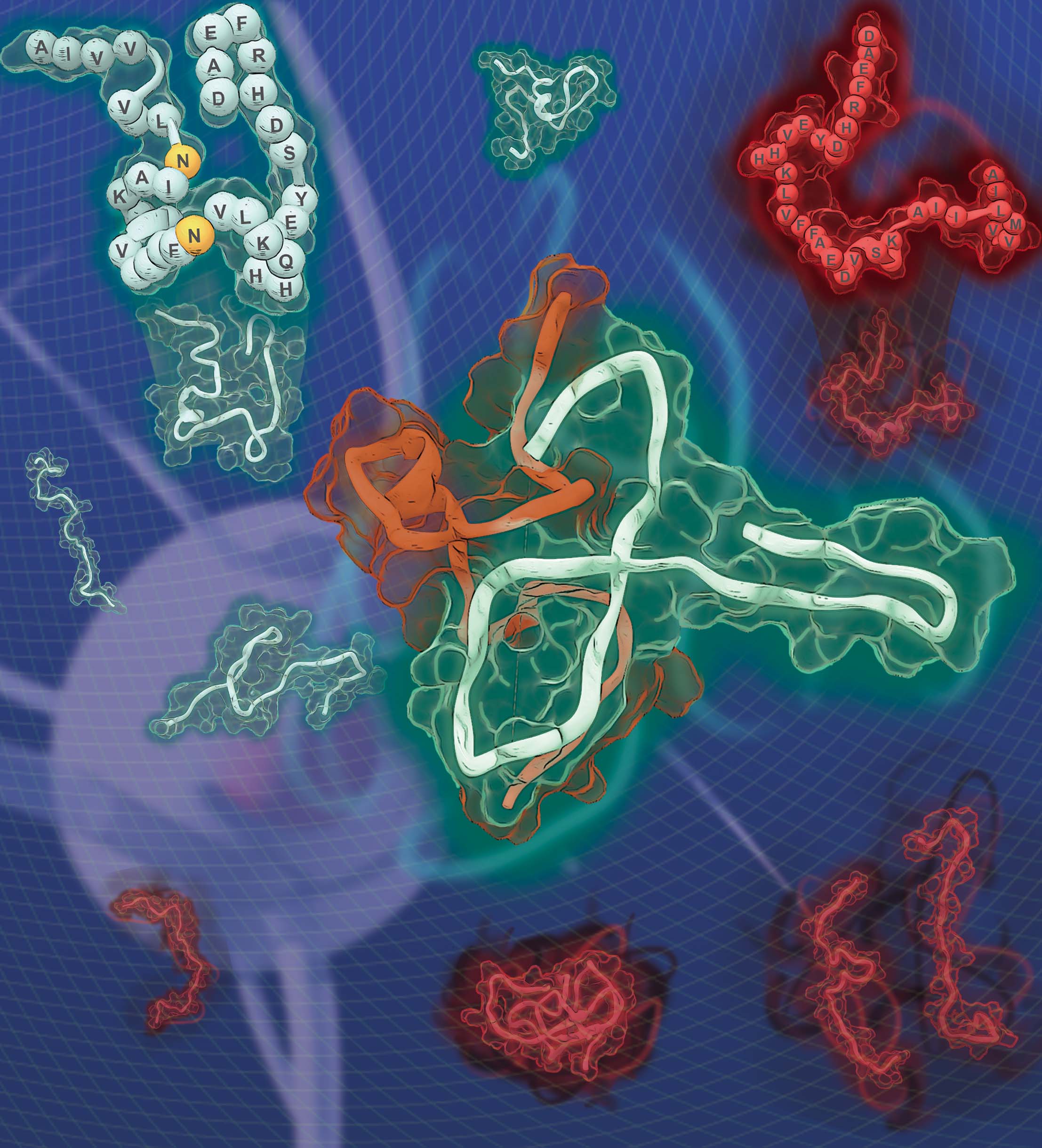
Kinetic Modulation of Amyloid-β (1-42) Aggegation and Toxicity by Structure-Based Rational Design
| author | Hugh I. Kim |
|---|---|
| Homepage | https://www.hughkimlab.com/ |
| journal | Journal of the American Chemical Society |
Several point mutations can modulate protein structure and dynamics, leading to different natures. Especially in the case of amyloidogenic proteins closely related to neurodegenerative diseases, structural changes originating from point mutations can affect fibrillation kinetics. Herein, we rationally designed mutant candidates to inhibit the fibrillation process of amyloid-β with its point mutants through multistep in silico analyses. Our results showed that the designed mutants induced kinetic self-assembly suppression and reduced the toxicity of the aggregate. A multidisciplinary biophysical approach with small-angle X-ray scattering, ion mobility-mass spectrometry, mass spectrometry, and additional in silico experiments was performed to reveal the structural basis associated with the inhibition of fibril formation. The structure-based design of the mutants with suppressed self-assembly performed in this study could provide a different perspective for modulating amyloid aggregation based on the structural understanding of the intrinsically disordered proteins.
https://pubs.acs.org/doi/10.1021/jacs.1c10173

Lim Dongjun
(First author, integrated course student)
Articles
-
 Chemical Fields: Directing Atom Migration in the Multiphasic Nanocrystal
Chemical Fields: Directing Atom Migration in the Multiphasic Nanocrystal
-
 Electronic Mechanism of In Situ Inversion of Rectification Polarity in Supramolec...
Electronic Mechanism of In Situ Inversion of Rectification Polarity in Supramolec...
-
 Direct observation of protein structural transitions through entire amyloid aggre...
Direct observation of protein structural transitions through entire amyloid aggre...
-
 Harnessing GLUT1-Targeted Pro-oxidant Ascorbate for Synergistic Phototherapeutics
Harnessing GLUT1-Targeted Pro-oxidant Ascorbate for Synergistic Phototherapeutics
-
 DNA-Damage-Response-Targeting Mitochondri-Activated Multifunctional Prodrug Strat...
DNA-Damage-Response-Targeting Mitochondri-Activated Multifunctional Prodrug Strat...
-
 Midwavelength Infrared Colloidal Nanowire Laser
Midwavelength Infrared Colloidal Nanowire Laser
-
 Kinetic Modulation of Amyloid-β (1-42) Aggegation and Toxicity by Structure-Based...
Kinetic Modulation of Amyloid-β (1-42) Aggegation and Toxicity by Structure-Based...
-
 Safeguarding the RuO2 phase against lattice oxygen oxidation during acidic water ...
Safeguarding the RuO2 phase against lattice oxygen oxidation during acidic water ...
-
 Mn-dopant differentiating the Ru and Ir oxidation states in catalytic oxides towa...
Mn-dopant differentiating the Ru and Ir oxidation states in catalytic oxides towa...
-
 Dynamic Water Promotes Lithium-Ion Transport in Superconcentrated and Eutectic Aq...
Dynamic Water Promotes Lithium-Ion Transport in Superconcentrated and Eutectic Aq...
-
 Isomeric sp2-C conjugated Porous Organic Polymer-mediated Photo- and Sono-catalyt...
Isomeric sp2-C conjugated Porous Organic Polymer-mediated Photo- and Sono-catalyt...
-
 Vertical-crystalline Fe-doped β-Ni oxyhydroxides for highly active and stable oxy...
Vertical-crystalline Fe-doped β-Ni oxyhydroxides for highly active and stable oxy...
-
 Extended Short-Wavelength Infrared Photoluminescence and Photocurrent of Nonstoic...
Extended Short-Wavelength Infrared Photoluminescence and Photocurrent of Nonstoic...
-
 Enhanced Thermopower of Saturated Molecules by Noncovalent Anchor-Induced Electro...
Enhanced Thermopower of Saturated Molecules by Noncovalent Anchor-Induced Electro...
-
 Mechanical Force for the Transformation of Aziridine into Imine
Mechanical Force for the Transformation of Aziridine into Imine
-
 A Small Molecule Strategy for Targeting Cancer Stem Cells in Hypoxic Microenviron...
A Small Molecule Strategy for Targeting Cancer Stem Cells in Hypoxic Microenviron...
-
 Mitochondria-targeted nanotheranostic: Harnessing single-laser-activated dual pho...
Mitochondria-targeted nanotheranostic: Harnessing single-laser-activated dual pho...
-
 Pb3Pb Nanosponges for Selective Conversion of Furfural to Furfuryl Alcohol under ...
Pb3Pb Nanosponges for Selective Conversion of Furfural to Furfuryl Alcohol under ...
-
 Deep Learning Optical Spectroscopy Based on Experimental Database: Potential Appl...
Deep Learning Optical Spectroscopy Based on Experimental Database: Potential Appl...
-
 Wettability of graphene and interfacial water structure
Wettability of graphene and interfacial water structure
Designed by sketchbooks.co.kr / sketchbook5 board skin